Radiomics May Predict Tumor Mutations in Advanced Stage Colorectal Cancer
New research examines prediction of five-year overall survival rate

Radiomics — an emerging field in medical imaging — shows promising results in tumor classification in colorectal cancer, new Radiology: Imaging Cancer research shows.
Radiomics uses data-characterization algorithms to extract a plethora of features from medical images. These radiomic features use inter-pixel relationships that can be used with machine learning (ML) to create artificial intelligence models. These models will eventually be able to accurately predict a number of disease characteristics.
“Radiomics is the process of extracting and analyzing huge amounts of quantifiable data from an image — data that cannot be seen by the naked eye,” said Adrian Negreros-Osuna, MD, a professor of radiology and coordinator of the 3D Printing Lab at Hospital Universitario, Monterrey, Mexico. “In many ways, radiomics is the future of radiology.”
Radiomics is primarily used in oncology as a quantitative tool for relaying information about tumor phenotype and clinical and genotypic end points. For example, preliminary studies have demonstrated that CT radiomic features correlate with clinical outcomes in esophageal cancer, tumor grade in melanoma, tumor histologic findings in renal cell carcinoma, tumor hypoxia, and angiogenesis in non-small-cell lung cancer.
“These studies demonstrate how radiomics can already be used to analyze massive amounts of quantifiable data and make accurate, clinically-significant predictions,” Dr. Negreros-Osuna said. “In the future, radiomics will have advanced to the point that we will be able to conduct virtual biopsies to predict things like genetics and diagnostics.”
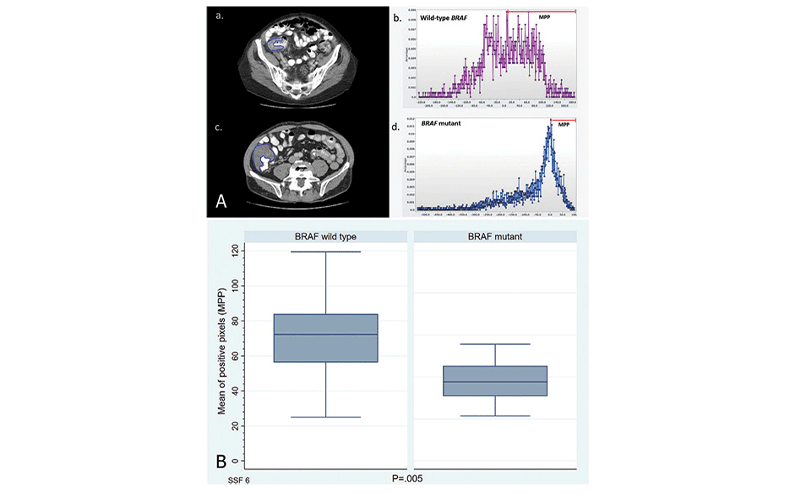
A, Histogram comparison shows significant differences in shape and in the mean of positive pixels (MPP; range highlighted in red) between (a., b.) wild-type BRAF and (c., d.) BRAF mutant colorectal tumors in the right colon. These tumors look reasonably similar on (a., c.) axial CT images. Blue outlines on CT images indicate colorectal tumors. B, Boxplot shows the difference in the MPP between wild-type BRAF and BRAF mutant tumors at a spatial scaling factor of 6 (SSF 6).
Negreros-Osuna, et al, Radiology: Imaging Cancer 2021; 298:3–17 ©️RSNA 2020
Accurately Detecting BRAF Mutation
In their Radiology: Imaging Cancer study, Dr. Negreros-Osuna and colleagues expanded the use of radiomics to colorectal cancer. The retrospective study, which included 145 patients with stage IV colorectal cancer, explores the potential of using such radiomic features as mean, standard deviation (SD), mean value of positive pixels (MPP), skewness, kurtosis, and entropy to detect the presence of BRAF mutation which is associated with poor prognosis and used to predict fiveyear overall survival.
“On the one hand, our study confirmed previous findings that patients who show less tumor texture heterogeneity have a lower survival rate,” Dr. Negreros-Osuna said. “On the other hand, our study went one step further and looked to see whether radiomics can be used to predict tumor mutation.”
Using only two radiomics features (SD and MPP), researchers were able to do exactly that. Specifically, they found that low grades of SD and MPP are indicative of the BRAF mutation.
“If you look at the imaging with the naked eye, you wouldn’t be able to differentiate the BRAF mutant from a non-BRAF mutant,” Dr. Negreros-Osuna explained. “But if you look at the radiomics’ histogram, one immediately sees that these two features show a statistically significant difference.”
These findings suggest that tumors with the BRAF mutation have lower levels of hypoxia and more vessels, which is why they do not respond to traditional therapy. For the nearly 10% of colorectal cancer patients who exhibit this mutation, this finding could mean getting the targeted treatment they need quicker for a potentially better prognosis.
“Diagnosing this mutation currently requires genetic profiling, which is both time-consuming and not particularly accurate,” Dr. Negreros-Osuna said. “Our study showed that radiomics can be used to accurately detect the BRAF mutation without having to do a biopsy, which is not only invasive, but also prone to sampling errors and often fails to represent the complete tumor heterogeneity.”
The study also demonstrated how skewness, a measure of symmetry, can be used to predict patient survival.
“All the patients in our study had stage IV colorectal cancer, meaning it is generally assumed that they would all have a similar rate of survival,” Dr. Negreros-Osuna said. “But with radiomics, we were actually able to predict who was going to survive longer.”
According to the study, patients with lower skewness survive 71 weeks, compared to those that have higher values, who only survive 40 weeks. Although the colorectal cancer study has significantly advanced the use and potential of radiomics, Dr. Negreros-Osuna says investigators are just beginning to scratch the surface.
“With every study, we gather new data on radiomics, which allows us to build better algorithms able to do increasingly complex tasks like predict genetic mutations, rate of survival and much, much more,” Dr. Negreros-Osuna said. “Radiomics has the potential to transform radiology and improve patient care, which is why it is so important that we keep doing this type of research.”
In addition to ongoing research, he also stresses the need for inter-institutional collaboration and data sharing.
“The more high-quality data we have, the quicker we’ll be able to build algorithms that can provide the same accurate results no matter where they are used,” he concluded. “If we do this, then in 15 or 20 years, using region of interest imaging to get the probability of genetic mutation with over 99% accuracy will be standard practice.”
For More Information
Access the Radiology: Imaging Cancer study, “Radiomics Texture Features in Advanced Colorectal Cancer: Correlation with BRAF Mutation and 5-year Overall Survival.”
Read the RSNA Position Statement on Colon Cancer Screening.
Read previous RSNA News stories on colorectal cancer: