Deep Brain Stimulation Shows Promise for Treatment-Resistant Depression
Imaging is critical to the revolutionary technique targeting specific areas of the brain



A groundbreaking technology now used to treat a range of neurological diseases also shows promise for treating depression in patients who have not responded to other therapies.
The concept of deep brain stimulation (DBS), which has been around for decades, has been approved by the U.S. Food and Drug Administration (FDA) to treat essential tremor, Parkinson’s disease, epilepsy and obsessive-compulsive disorder. Deep brain stimulation is not yet approved to treat depression.
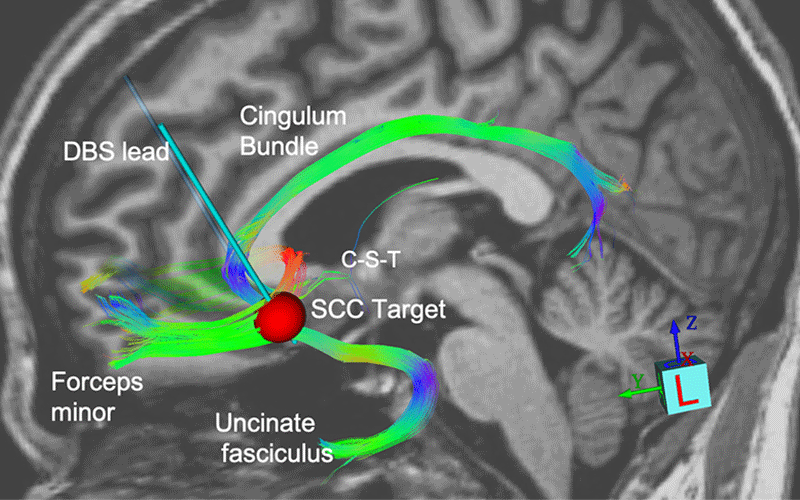
The DBS technique involves placing implanted electrodes deep in the brain to target specific areas responsible for the symptoms of each disorder, said researcher Helen Mayberg, PhD, professor of neurology, neurosurgery, psychiatry, and neuroscience, and founding director of the Nash Family Center for Advanced Circuit Therapeutics at the Icahn School of Medicine at Mount Sinai, NY, who pioneered DBS for depression and is leading new research efforts.
Imaging is critical to the success of the DBS technique, Dr. Mayberg said.
“Obviously, inserting something in the brain requires that we know where to put it,” Dr. Mayberg said. “And imaging, even for just the basics of high-resolution anatomy, is essential. With DBS and Parkinson’s disease, imaging has been a critical variable in improving the precision of the implanted electrodes.”
In recent research in the American Journal of Psychiatry, Dr. Mayberg and colleagues demonstrated that implanting paired electrodes into a region of the brain located in the subcallosal cingulate cortex (Area 25) could have an anti-depressive effect sustained over a long period of time in patients with treatment-resistant depression.
“Imaging is critical in cases where we know we are at the convergence zone of many white matter bundles and that small differences in anatomy end up impacting very different, remote parts of the brain.”
HELEN MAYBERG, PHD
Dr. Mayberg’s study documents outcome data over four to eight years for 28 patients who underwent DBS of the subcallosal cingulate for treatment-resistant depression.
Response and remission rates were maintained at ≥50% and ≥30%, respectively, from two to eight years of follow-up. In addition, three-quarters of participants met treatment response criterion for more than half of the time they participated in the study, with 21% of patients demonstrating continuous response to treatment from year one. Because depression has no definitive pathology, the idea behind DBS was based on results from earlier research using functional imaging, Dr. Mayberg said.
“Imaging findings became the impetus and the rationale for the target itself, at least. in our experiments,” she said.
The focus on Area 25 was logical, Dr. Mayberg said, because of its connection to negative mood. “This area becomes overactive with induction of negative mood states, which we could see in blood flow PET scans,” Dr. Mayberg said. “Its activity was attenuated when patients were successfully treated for depression. We saw the signal change with glucose metabolism and blood flow. Functional imaging led us to the target and helped us turn down the activity directly with focal electrical stimulation using the implanted electrode.”
Imaging not only became the conduit to performing the implants but also helped in the understanding of their impact on the location the implants were affecting.
“Imaging is critical in cases where we know we are at the convergence zone of many white matter bundles and that small differences in anatomy end up impacting very different, remote parts of the brain,” Dr. Mayberg said.
Identifying individuals suffering from depression who will benefit from specific therapies, as well as developing innovative treatments like DBS, requires a better understanding of the biology of depression.
MRI Assesses Blood Brain Barrier
One area that has been implicated in major depressive disorder is disruption of the blood brain barrier (BBB).
Research led by Kenneth Wengler, PhD, and including Kwan Chen, MD, both from the radiology department at State University of New York at Stony Brook, investigated differences in BBB integrity between patients with major depressive disorder and healthy subjects using the recently developed Intrinsic Diffusivity Encoding of Arterial Labeled Spins (IDEALS) MRI technique.
“This technique basically lets us assess the permeability of water through the blood brain barrier,” Dr. Chen said. “We magnetically label the water in arteries and observe how it goes into the brain and where it flows. But with this technique, we are also able to tease out a little more than that — we are able to see what goes through the barrier itself.”
In this small trial of 28 patients, researchers observed lower water movement through the BBB. “This finding basically goes against the conventional idea of blood brain barrier damage, since damage to the blood brain barrier would likely result in more leaking.”
“This finding looks at BBB damage in a different way, since the traditional view of BBB damage would result in more leaking,” Dr. Chen said. “One possibility is that the pumps that regulate water movement in the BBB aren’t working due to intracellular metabolic changes.”
For More Information
Access the study, “Long-Term Outcomes of Subcallosal Cingulate Deep Brain Stimulation for Treatment-Resistant Depression,” at AJP.psychiatryonline.org.