Medical Imaging Data Resource Center (MIDRC)
RSNA is collaborating with radiology organizations around the world to curate a comprehensive repository of medical imaging data for research.
The Medical Imaging and Data Resource Center (MIDRC), funded through the National Institute of Biomedical Imaging and Bioengineering (NIBIB) under the National Institutes of Health (NIH), is an open-access platform for medical images and associated data.

About MIDRC
Hosted by the University of Chicago, MIDRC is a collaborative effort between RSNA, the American College of Radiology (ACR) and the American Association of Physicists in Medicine (AAPM).
RSNA’s contribution to MIDRC includes the RSNA International COVID-19 Open Radiology Database (RICORD), a substantial body of imaging data with annotations and supporting clinical information for use in education and research.
MIDRC data collection
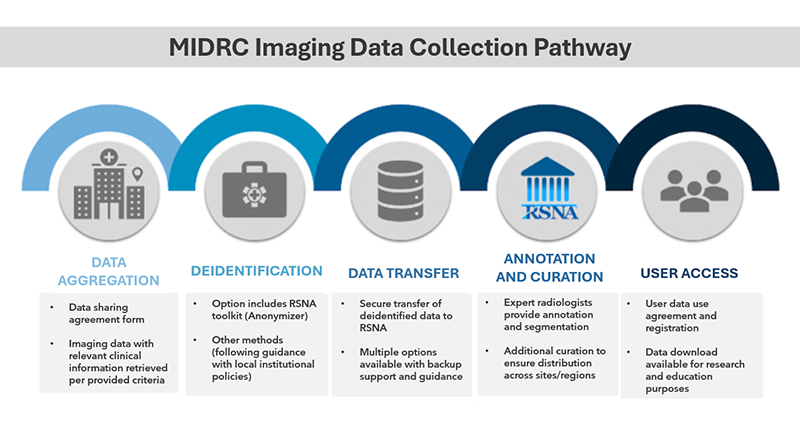
RSNA is collecting and publishing data through MIDRC and has assembled an international task force of scientists and radiologists to support this effort.
RSNA has developed data selection criteria, data sharing agreements and tools to help sites organize, de-identify and transfer data. The RSNA-MIDRC data collection pathway will enable radiology organizations to contribute data to MIDRC safely and conveniently.
Assembling broad and diverse datasets is essential to supporting quality research leading to improvements in diagnosis care.
How to participate
Is your organization interested in contributing data for research to MIDRC? RSNA invites you to complete the MIDRC data intake form to express your interest.
What’s next?
We are continually expanding MIDRC's scope to include more types of medical data, supporting research that drives advancements in artificial intelligence. Stay tuned for further updates as MIDRC continues to evolve.
MIDRC seminar series
Attend the MIDRC seminar series to gain insights directly from the MIDRC team! Seminars occur on the third Tuesday of the month and include live Q&A sessions.
Learn more and register today.
MIDRC newsletter
Looking to stay informed on the latest MIDRC developments? Join the mailing list and receive updates on MIDRC’s progress.
Contact us
We welcome your questions and suggestions, which can be sent to informatics@rsna.org.